
Renewable solutions
6 minute read
The problem isn’t carbon itself, but rather its source: how renewable carbon combats climate change
It’s a fact that if you measure greenhouse gas emissions at the tailpipe of a vehicle, you’ll get roughly the same results regardless of whether the engine runs on fossil-based fuels or on renewable fuels produced from bio-based raw materials. Why, then, are renewable fuels advertised as creating significantly less emissions? And what is renewable carbon? If you’ve ever found that confusing, then this article is for you.
Making sense of the answer requires us to have a closer look at what we call the “carbon cycle” – or the natural circulation of carbon between living beings and different parts of the planet. Here are some examples of how that works (and yes, you might remember some of these terms from your science class – if you weren’t writing sticky notes to your friends then):
Photosynthesis – plants absorb carbon (in the form of carbon dioxide, i.e. CO2) from the atmosphere and use it to grow
Respiration – animals release carbon (in the form of CO2) into the atmosphere when breathing
Diffusion/Exsolution – oceans exchange carbon (CO2) with the air above them
These interactions between three carbon reservoirs – the atmosphere (air), the biosphere (living things) and the hydrosphere (oceans) – form a large part of Earth’s carbon cycle. This carbon cycle is delicately balanced. However, there is another very important reservoir: the lithosphere. The lithosphere is the uppermost, solid part of the Earth. It stores large amounts of carbon, mostly in the form of coal, gas and oil. When “trapped” in the lithosphere, carbon stays there for very, very long periods of time – even for millions of years. Left untouched, it is only released very occasionally through events such as volcanic eruptions, leading to venting carbon into the atmosphere in the form of CO2.
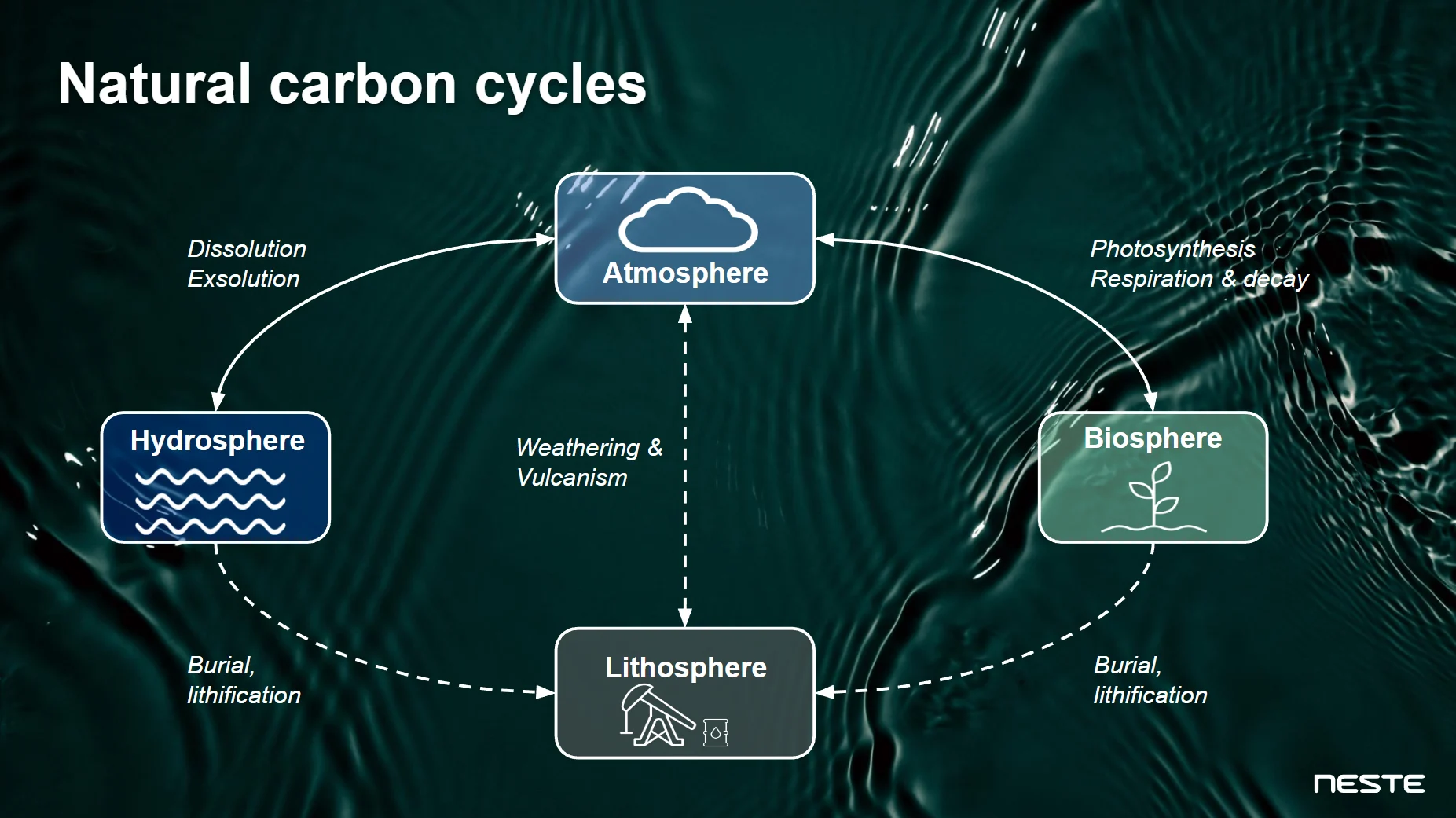
Earth’s carbon cycles (simplified).
While movement of carbon between the biosphere, atmosphere and hydrosphere can be thought of as part of the natural short-term carbon cycle, movement of carbon into and out of the lithosphere can be understood as part of the long-term carbon cycle. Thus, what we call renewable carbon comes from the biosphere, atmosphere and hydrosphere – but not from the lithosphere.
And there’s the problem.
For the past few centuries, lithospheric carbon has not remained untouched, as the human species has increasingly taken advantage of it in the course of industrialization. In fact, we’ve been mining ever-increasing amounts of coal, gas and oil for example. We have been converting these deposits of carbon in the lithosphere to energy in the form of heat, electricity and fuels (mostly by burning it) and allowing the resulting emissions to permeate the Earth’s atmosphere. This massive disruption of the planet’s natural carbon cycles has led to a drastic increase in the concentration of CO2 in the atmosphere, creating the greenhouse effect, warming the planet and causing the global climate to undergo significant changes. The problem isn’t that CO2 can be found in the atmosphere – the problem is that the concentration of CO2 in the atmosphere has increased by almost 50 percent since the beginning of the Industrial Age.
Good CO2 and bad CO2?
Considering the natural order of things, CO2 isn’t evil as such. It’s just a chemical compound – and a very important one. Without CO2, plants wouldn’t grow and life would not be possible. However, when considering the effects of human activity on the Earth’s carbon cycles, it becomes clear that the source of the carbon or CO2 matters a lot. So, in a sense, we could say that CO2 is bad if the carbon is coming from the lithosphere – also known as “fossil” CO2. It’s the result of taking carbon from the long-term cycle and adding it to the short-term cycle. By burning increasingly large amounts of fossil resources, we have added huge amounts of extra carbon from the lithosphere to the atmosphere. The result: We increased the total CO2 concentration in the atmosphere, because the short-term cycle doesn’t have a natural way of binding that carbon, or getting it back below the Earth’s surface fast enough.
Hence, it’s safe to say that we need to stop (or at least drastically cut down) our use of fossil resources if we want to limit global warming. We need to let the long-term cycle do its job and hang onto its share of carbon.
This brings us to the “good” CO2: using carbon that is already part of the short-term cycle and that is naturally circulating between the biosphere and atmosphere.
Imagine there were no humans on Earth. There would still be carbon moving from the biosphere to the atmosphere and vice versa as plants would be growing and decaying – and there wouldn’t be an issue with that. This is the carbon cycle we can make use of. Before the plant decays and emits its carbon as CO2, we take this carbon on a little detour and turn it into the biofuel mentioned at the beginning of this article. Why not delay that inevitable moment of emissions a tiny bit? The overall concentration of carbon in the atmosphere isn’t affected by that.
The goal of carbon neutrality
The more we develop and use technologies that accomplish the above, the closer we get to carbon neutrality – which means not adding any more carbon to the short-term carbon cycle than what can be naturally bound.
Today, of course, fossil resources are still used for many things, including in the conversion to energy. Fossil-based energy may still be used in the production of renewable and bio-based fuels or manufacturing of materials such as plastics: the truck delivering the raw materials may run on fossil fuel, for example, or the processing plant may be powered by energy from coal or gas.
This is why you may see reports that renewable products lead to maybe 70, 80 or 90 percent less emissions than their fossil-based counterparts. The remaining emissions can be reduced further, if all fossil energy sources are replaced with renewable ones or if any remaining emissions are captured, for example.

The difference between renewable and fossil carbon.
While we think it will be possible to have actual carbon neutrality (or even negativity if we manage to take more carbon from the atmosphere than we add to it!) in the future, there is already the potential to create a huge difference with the solutions available today – right now and at scale. And since every kilogram of fossil carbon that we can avoid from being released into the atmosphere counts, we would be well advised to make use of these renewable solutions to replace bad CO2 with good CO2. Bottom line: Yes, an engine running on renewable fuels also emits CO2, but it’s the good one.
Credits: Neste